 
Salad and a bowl of cereal are other examples of a mixture with distinct parts. For example, rocks, twigs, insects and leaves are visible components of soil. In some mixtures, each of the components can be seen. Since substances that form mixtures are not bonded together mixtures are not pure substances. In a mixture, each substance keeps its individual properties and identity. Solids, liquids or gases can be combined to form mixtures. A mixture is a combination of two or more substances that are not chemically combined. Elements can combine in different ways to form other substances called compounds and mixtures. Most substances or matter are not made of just one element. Iron is a metal that reacts easily with water forming rust, while aluminum does not. Hydrogen is another element that is highly reactive and it causes explosions when it comes in contact with certain elements. Oxygen is highly reactive and it readily combines with other substances during burning. Hydrogen and oxygen are nonmetals that are similar in that they are both colorless gases however, each gas has distinct characteristic properties.Įlements also have different reactivity, which describes there ability to form chemical bonds with other substances. Diamond is transparent and the hardest material found in nature. One form, graphite, is a soft grey substance that makes up pencil lead.Īnother form that looks very different from graphite is diamond. Other elements, such as carbon (C), hydrogen (H), oxygen (O) and chlorine (Cl), are classified as nonmetal.Ĭarbon is a nonmetal that is a solid at room temperature, but exists in several different forms (graphite, diamond, and coal), each of which has distinct properties. 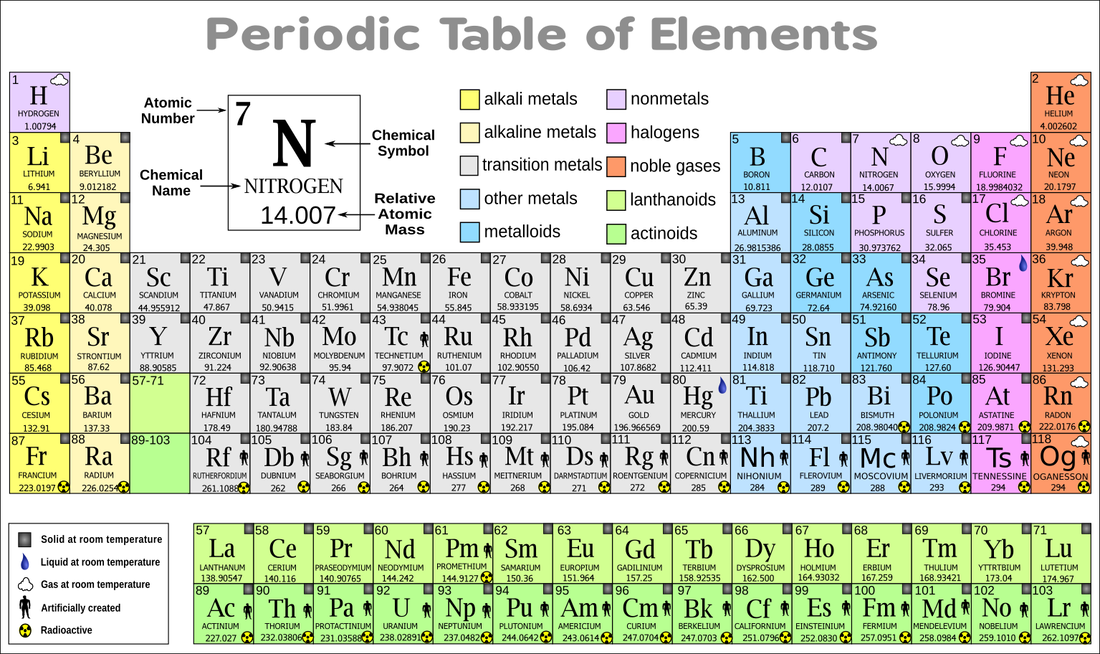
For example, sodium (Na) is a light, soft metal that is nonmagnetic, while iron is a magnetic metal that is denser than sodium and aluminum. Individual metallic elements have distinct characteristic properties such as malleability, ductility, and relatively high density. Iron is silvery grey and heavy, while aluminum is silvery white and lightweight in comparison. Iron and aluminum are two shinny metals, but they differ slightly in their properties. Some elements, such as iron (Fe) and aluminum (Al) are classified as metals. Properties such as magnetic attraction, conductivity, density, pH, boiling point and solubility are characteristic properties that can be used to identify substances. The Periodic Table of Elements is used to organize elements based on properties such as their reactivity, state of matter, conductivity or density.Īll matter has a variety of properties, some of which are characteristic of the substance. Characteristic properties do not depend on the amount of the substance. The structure of the atoms determine an elements’ properties. For example, carbon is represented by C, oxygen by O, and He for helium.Įach element also has different properties or characteristics. 
This letter symbol is typically made up of one or two letters. Atoms are made up of different numbers of subatomic particles (neutrons, protons, and electrons).Įlements are represented by their chemical symbol on the Periodic Table of Elements. The main differences between atoms are the structure of the atom that makes it up. Each element is made up of atoms that differ from other elements. The remaining elements are synthetic and not found naturally instead they are made in the laboratory.Īll matter is made of different combinations of about 100 pure substances called elements.Īn atom is the smallest particle of an element that has all the properties of that element. Of these elements, 92 are naturally found in nature such as carbon, oxygen, gold, silver, and iron. Atoms are too small to be seen without special magnification.įor example, a gold ring can be broken into smaller and smaller pieces until the pieces are no longer visible. An element is a pure substance made up only of a single type of atom. Matter is made of particles called atoms and molecules.Ītoms are the basic building blocks of most matter around us. The classification of the state of matter is based on observable characteristic properties. For instance, the land we walk on is solid, oceans and rain are liquid, and the air we breathe is a mixture of gases. Often we can determine the state of matter by observing the substance with the naked eye. 
Most matter on Earth exists in three states: solid, liquid, and gas.Įxamples of solids, liquids, and gases can be found all around us. The term phase can also be used to describe which state matter is in. The physical form in which matter exists is called a state of matter. Matter is anything that has mass and volume. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
